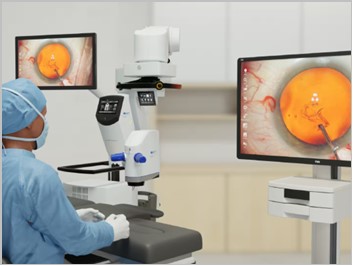
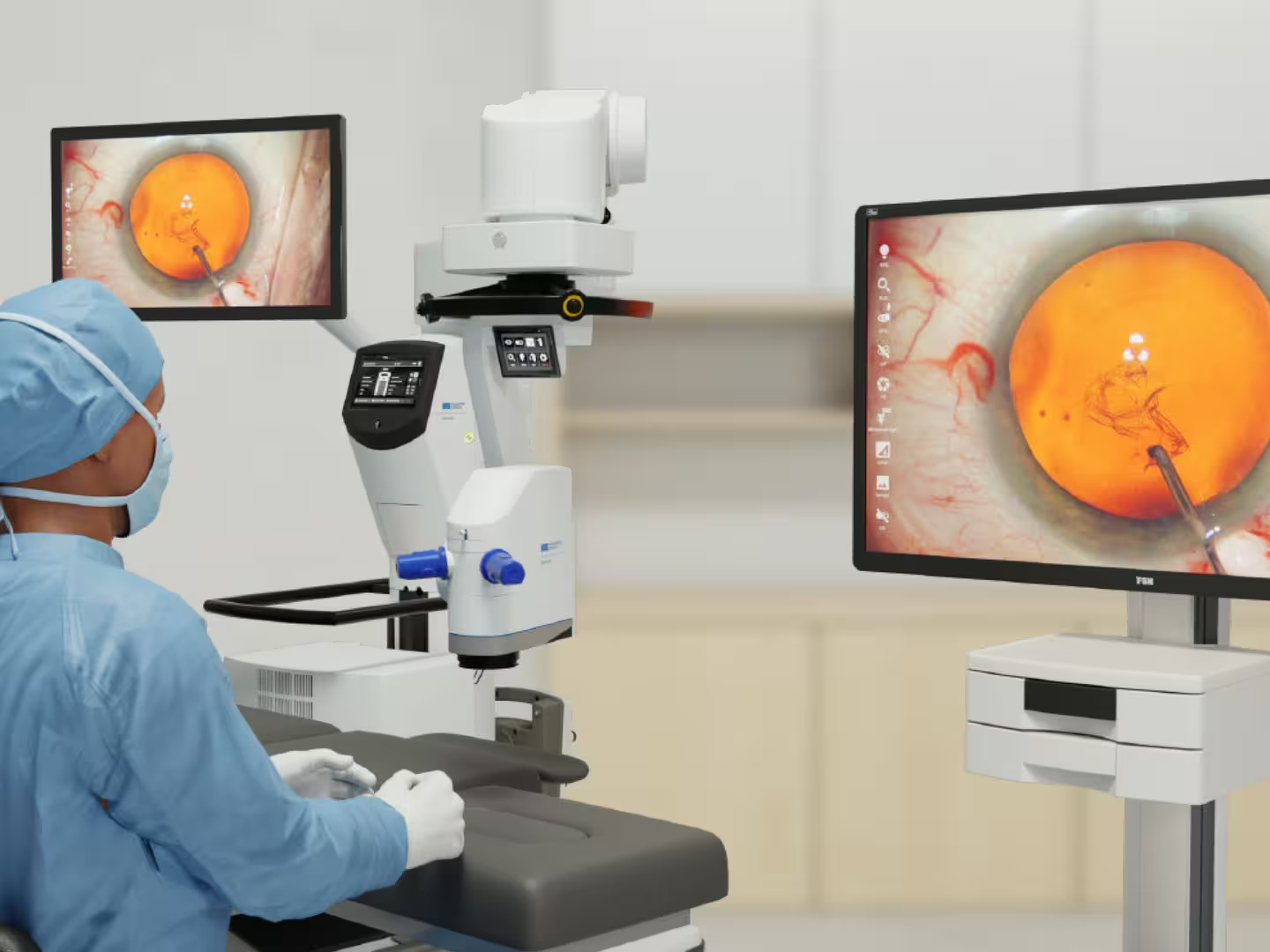
More News
Want to Read Locked Articles?
Already Have an Account?
Register A Corporate Account
A corporate account gives you access to licensed reports and subscriptions, the latest news, a personalized dashboard, and weekly emails with news and data.